Accelerating Drug Design for Ovarian and Prostate Cancer
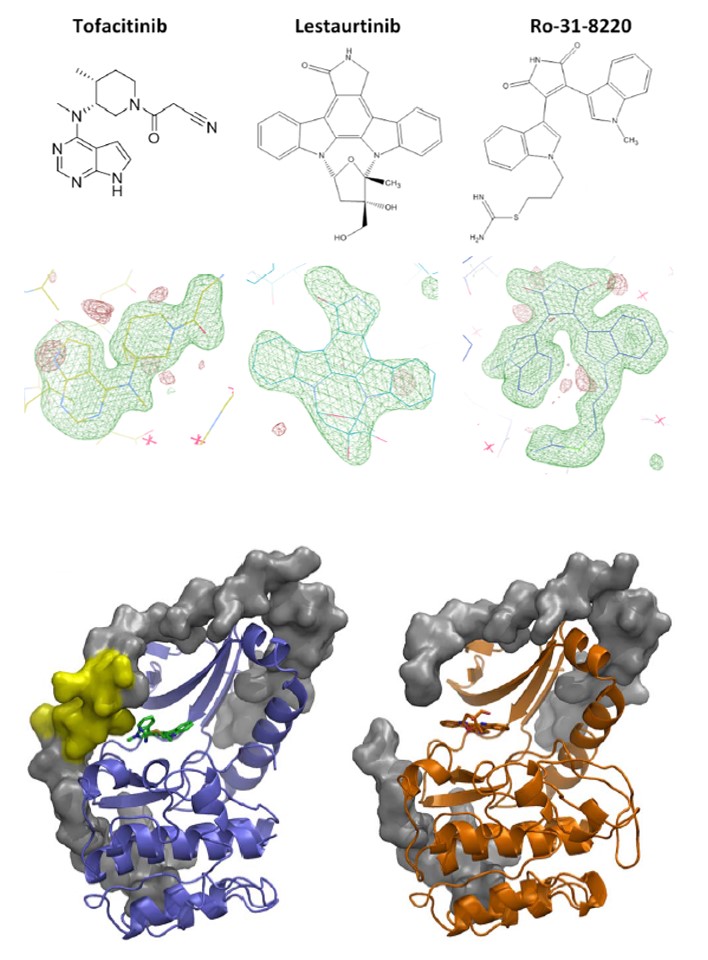
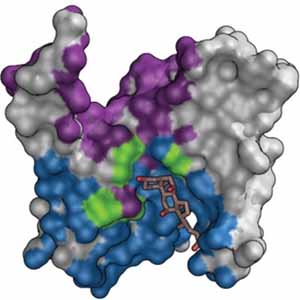
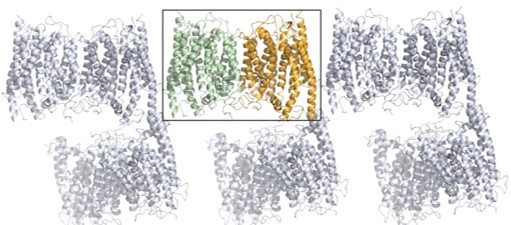
Cancer cells often overexpress and release certain proteins that help the cancer spread. The protein PRK1 is one such protein expressed in prostate and ovarian cancer. Because of this, PRK1 is a target for therapeutics for these cancers, and indeed several inhibitors to PRK1 are in clinical trials. This study by researchers at Celgene, using crystal structures solved at beamlines 5.0.1, 5.0.2, and 5.0.3, elucidated the structure of PRK1 both with and without these inhibitors, showing some interesting structural features that will accelerate the design of even more potent and selective drugs to target PRK1.
P. Chamberlain, S. Delker, B. Pagarigan, A. Mahmoudi, P. Jackson, M. Abbasian, J. Muir, N. Raheja, B. Cathers, “Crystal Structures of PRK 1 in Complex with the Clinical Compounds Lestaurtinib and Tofacitinib Reveal Ligand Induced Conformational Changes,” PLOS ONE August (2014).
Targeting the Kinase
Protein kinases are used in a huge number of cellular processes. By catalyzing the transfer of a phosphate group, they control cell signalling and influence cell proliferation, cell adhesion, and survival, among a number of other physiological important processes. One drug that has been developed to target kinases is the cancer drug Ruxolitinib, which is a kinase inhibitor and therefore can limit cell proliferation. It has been approved for the treatment of the myeloproliferative neoplasms. But how does it work? In this study, scientists determined the crystal structure of a kinase domain in complex of Ruxolitinib at a high enough resolution to show some of the exact chemical changes that the drug induces. In addition, the study indicates how the drug can me modified to be even more effective.
Y. Duan, L. Chen, Y. Chen, X.G. Fan, “c-Src Binds to the Cancer Drug Ruxolitinib with an Active Conformation,” PLOS ONE Sept 8 (2014).
Why Some Cells Should Die
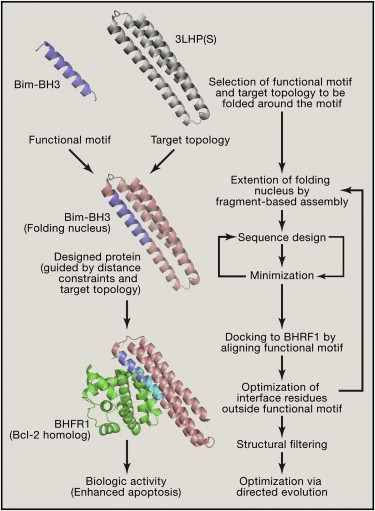
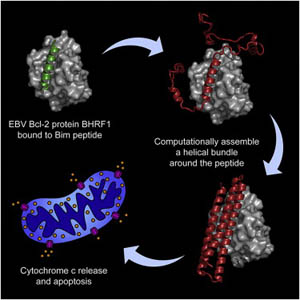
Cells go through a natural process called apoptosis when they are damaged or can no longer function properly. By taking themselves out of commission they reduce the danger to the organism when, for instance, they are infected with a virus. But this process of apoptosis is not advantageous for a virus, which infects a cell and then forces the cell to make more copies of itself, as is the case with the Epstein-Barr virus (EBV). The EBV actually produces inhibitor proteins to counteract the process of apoptosis, keeping the host cell alive long enough to infect other cells. The scientists in this study used a new process of de-novo protein design to computational build a novel protein that would bind to and inhibit the inhibitor. The method was successful in determining a new protein inhibitor of EBV, and so can now we used to design proteins to fight other infectious agents and cancer.
E. Procko, G.Y. Berguig, B.W. Shen, Y. Song, S. Frayo, A.J. Convertine, D. Margineantu, G. Booth, B.E. Correia, Y. Cheng, W.R. Shief, D.M. Hockenbery, O.W. Press, B.L. Stoddard, P.S. Stayton, D. Baker, “A computationally designed inhibitor of an Epstein-Barr viral Bcl-2 protein induces apoptosis in infected cells,” Cell 157, 1644-1656 (2014).
Stimulating Insulin Production in the Fight Against Type-II Diabetes
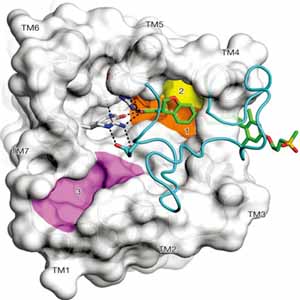
Treatments for type-2 diabetes have centered on targeting the human receptor protein GPR40, since it is a fatty-acid receptor that can enhance glucose-dependent insulin secretion. TAK-875 is a drug developed by the company Takeda to stimulate insulin secretion by binding to this receptor. Researchers at Takeda have released a publication showing the details of the mechanism of action of this drug for the first time, based on protein structures solved at beamline 5.0.3.
A. Srivastava, J. Yano, Y. Hirozane, G. Kefala, F. Gruswitz, G. Snell, W. Lane, A. Ivetac, K. Aertgeerts, J. Nguyen, A. Jennings, K. Okada, “High-resolution structure of the human GPR40 receptor bound to allosteric agonist TAK-875,” Nature 513, 124 (2014).
Keeping the Heart Pumping and the Neurons Firing
Nearly every cell in the human body contains ion channels; conduction of potassium across membranes is what helps keep muscles moving, the heart pumping, and brain neurons firing. And channel proteins also have to be selective: calcium channels selectively drive Ca2+ into cells despite a 70-fold higher extracellular concentration of Na+. These studies revealed the molecular mechanisms by which channel proteins are both selective and efficient.
L. Tang, T.M. G. E.-Din, J. Payandeh, G.Q. Martinez, T.M. Heard, T. Scheuer, N. Zheng, W. Catterall “Structureal basis for Ca2+ selectivity of a voltage-gated calcium channel,” Nature 505, 56 (2014).
How Cells Distinguish Between Young and Old
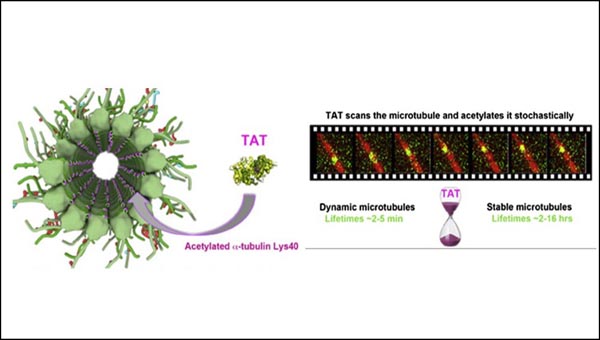
Researchers in this study used crystal structures solved at beamline 5.0.1 to show how the enzyme TAT distinguishes between slow and fast turnover rates of microtubules and marks them accordingly. The work is broad-reaching because marking of microtubules affects cell division, mobility, and lifetime.
A. Szyk, A.M. Deaconescu, J. Spector, B. Goodman, M.L. Valenstein, N.E. Ziolkowska, V. Kormendi, N. Grigorieff , A. Roll-Mecak, “Molecular basis for age-dependent microtubule acetylation by tubulin acetyltransferase,” Cell 157, 1405 (2014).
Understanding the Virus-Antibody Arms Race
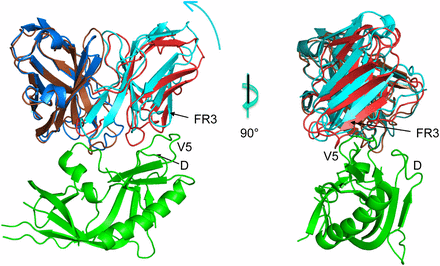
Rapidly evolving pathogens, such as human immunodeficiency and influenza viruses, escape immune defenses provided by most vaccine-induced antibodies. This works provides a overview of the structural mechanisms underpinning the virus-antibody arms race.
D. Fera, A.G. Schmidt, B.F. Haynes, F. Gao, H.X. Liao, T.B. Kepler, and S.C. Harrison, “Affinity maturation in an HIV broadly neutralizing B-cell lineage through reorientation of variable domains,” PNAS USA 111, 10275 (2014).
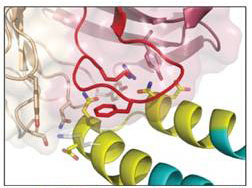
Cooperative Binding to Brain Receptor Proteins
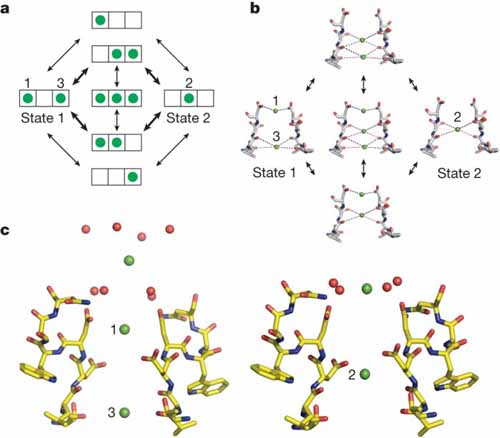
The nicotinic acetylcholine receptor (nAChR) and the acetylcholine binding protein (AChBP) are neuron receptor proteins that signal for muscular contraction upon a chemical stimulus. This study shows for the first time cooperative binding of certain ligands, showing interactions between various domains of this receptor.
K. Kaczanowska, M. Harel, Z. Radić, J.P. Changeux, M.G. Finn, and P. Taylor, “Structural basis for cooperative interactions of substituted 2-aminopyrimidines with the acetylcholine binding protein,” PNAS, 111 10749 (2014).
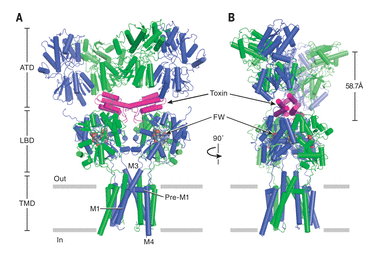
Toxins in the Brain
Membrane-embedded receptors in the human brain have myriad responsibilities in human health: pain sensing, learning and memory, nervous system development, and detection of injury. In a series of high-profile studies, these investigators solved the structures of many of these important proteins alone and in complex with toxins, giving critical insight into exactly how these proteins sense and respond to extracellular signals.
L. Chen, K. L. Duerr, E. Gouaux, “X-ray structures of AMPA receptor–cone snail toxin complexes illuminate activation mechanism,” Science, 345 1021 (2014).
The Dopamine Receptor Unravelled
This work show for the first time the structure and domain arrangement of the main dopamine receptor in the brain: “…one of the most important receptors in the brain — a receptor that allows us to learn and remember, and whose dysfunction is involved in a wide range of neurological diseases and conditions, including Alzheimer’s, Parkinson’s, schizophrenia and depression….”. from Nature News & Views: “Neuroscience: A structure to remember.”
C.-H. Lee, W. Lu, J.C. Michel, A. Gorhring, J. Du, X. Song, E. Gouaux, “NMDA receptor structures reveal subunit arrangement and pore architecture,” Nature 511, 191 (2014).
Stopping a Virus From Keeping Infected Cells Alive
The Epstein-Barr virus not only infects cells, but then blocks cell apoptosis, keeping the infected cells alive so they will go on to spread the infection. In this study, a novel protein inhibitor was designed computationally, then grown, crystallized and structurally determined at beamline 5.0.2, which revealed how the protein binds to the virus and allows apoptosis. The designed protein was then shown to suppress tumor growth and extend survival in animal models.
E. Procko, G.Y. Berguig, B.W. Shen, Y. Song, S. Frayo, A.J. Convertine, D. Margineautu, G. Booth, B.E. Correia, Y. Cheng, W. Schief, D.M. Hockenbery, O.W. Press, B. L. Stoddard, P.S. Stayton, D. Baker, “A Computationally Designed Inhibitor of an Epstein-Barr Viral Bcl-2 Protein Induces Apoptosis in Infected Cells,” Cell 157, 1644 (2014).
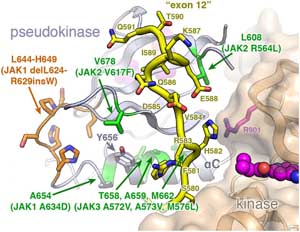
Kinase Activity and Cytokine Signalling
Cytokine signaling is essential for cell growth, hematopoiesis, and immune system function. Cytokines can induce dimerization of their receptors, which in turn induces activation of other proteins such as Janus kinases (JAKs) leading to further signalling inside the cell. In this study, researchers used crystal structures obtained at beamline 5.0.1 of certain JAK proteins to deduce a connection between cancer-associated mutations in the protein and unusual kinase activity.
P. J. Lupardus, M. Ultsch, H. Wallweber, P.B. Kohli, A.R. Johnson, C. Eigenbrot, “Structure of the pseudokinase–kinase domains from protein kinase TYK2 reveals a mechanism for Janus kinase (JAK) autoinhibition”, PNAS 111, 8025 (2014).
Transporters As Drug Targets
A class of transporters, the ASBT proteins, are under scrutiny as potential drug targets for treatment of hypercholesterolaemia and type 2 diabetes. These proteins are bile acid transporters could also be used for delivering drugs orally, and so their structure is of immense importance to medical research. This study, using structures solved at beamline 8.2.2, revealed some of the first three-dimensional images of these important transporters.
X. Zhou, E.J. Levin, Y. Pan, J.G. McCoy, R. Sharma, B. Kloss, R. Bruni, M. Quick, M. Zhou, “Structural basis of the alternating-access mechanism in a bile acid transporter,” Nature 505, 569 (2014).
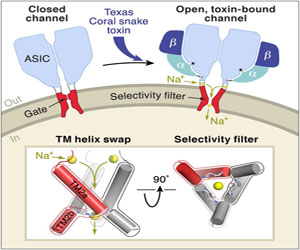
The Pain-Sensing Gatekeepers of the Cell
Certain ion channels can detect inflammation or injury by monitoring concentrations of protons outside the cell wall. Using beamline 5.0.2, Gouaux and coworkers solved the structure of one of these ion channels in complex with a pain-inducing toxin from the Texas Coral snake, revealing exactly how this protein responds to extracellular toxins.
I. Baconguis, C.J. Bohlen, A. Goehring, D. Julius, E. Gouaux, “X-Ray Structure of Acid-Sensing Ion Channel 1-Snake Toxin Complex Reveals Open State of a Na-Selective Channel,” Cell 156, 717 (2014).
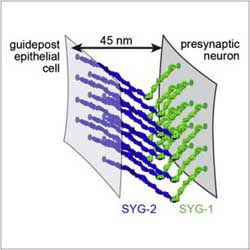
Architecture of Cell Adhesion
The ability of cells to “stick together” is what makes multi-ceullular life possible, and so cell adhesion molecules have evolved across all major species. The level of “stickiness” is regulated by interactions between cells. Crystal structures solved at beamlines 8.2.1 and 8.2.2, along with biochemical characterization have show the architecture of this adhesion, and have furthermore shown that the proteins involved may do more than just facilitate the interface between cells; they may also be used in intercellular signaling.
E. Ozkan, P.H. Chia, R.R. Wang, N. Goriatcheva, D. Borek, Z. Otwinowski, T. Walz, K. Shen, K.C. Garcia, Cell 156, 482. (2014).
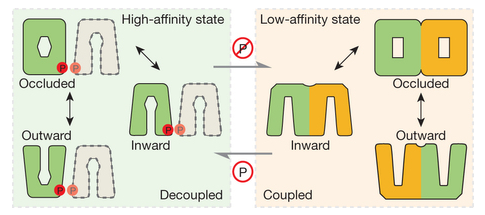
How Plants Deal With Varying Nitrate Levels
Plants require nitrate for proper growth, but the availability of nitrates in soil can vary by orders of magnitude. These studies, using structures obtained at beamline 8.2.1, revealed how a nitrate transporter is able to switch between two states for high or low affinity in order to transport the appropriate amount of nitrate to meet the needs of the plant.
J. Sun, J.R. Bankston, J. Payandeh, T.R. Hunds, W.N. Zagotta, N. Zheng, “Crystal structure of the plant dual-affinity nitrate transporter NRT1.1”, Nature 507, 73 (2014).
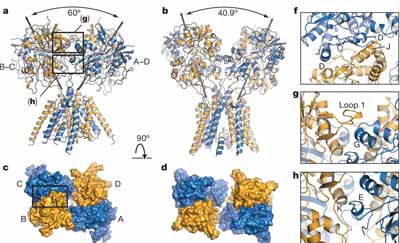
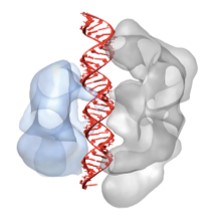
A Big Step Toward Targeted Gene Control
Bacteria use the CRISPR-Cas9 system to delete foreign DNA that comes from viruses. Now, humans are starting to realize its potential in targeted genome editing and gene regulation. The beauty of the Cas9 system is that the same enzyme can be used to target nearly any gene in the human genome. But how does it work? Doudna and coworkers used crystallographic structures from 8.2.1 and 8.2.2, and cryo-EM structures to determine two major Cas9 enzymes subtypes, and their orientation and binding to DNA.
M. Jinek, F. Jiang, D.W. Taylor, S.H. Sternberg, E. Kaya, E. Ma, C. Anders, M. Hauer, K. Zhou, S. Lin, M. Kaplan, A.T. Iavarone, E. Charpentier, E. Nogales, J.A. Doudna, “Structures of Cas9 Endonucleases Reveal RNA-Mediated Conformational Activation,” Science, 343 (2014).
Computationally Designing Drugs
This study used computational design to generate small, stable protein that accurately mimic the viral epitope structure and induce potent neutralizing antibodies. The results were validated using structures obtained at beamline 5.0.1. The results not only provide a promising leads for development of virus vaccines for human respiratory illnesses, but also provide proof of principle for a variety of other vaccine targets, such as human immunodeficiency virus and influenza.
B.E. Correia, J.T. Bates, R.J. Loomis, G.Baneyx, C. Carrico, J.G. Jardine, P. Rupert, C. Correnti, O. Kalyuzhniy, V. Vittal, M.J. Connell, E. Stevens, A. Schroeter, M. Chen, S. MacPherson, A.M. Serra, Y. Adachi, M.A. Holmes, Y. Li, R.E. Klevit, B.S. Graham, R.T. Wyatt, D. Baker, R.K. Strong, J.E. Crowe, P.R. Johnson and W.R. Schief, “ Proof of principle for epitope-focused vaccine design,” Nature, 507 201 (2014).
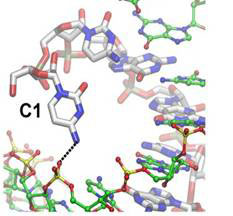
The Versatility of RNA
The replication of RNA without the aid of proteins is thought to have been a critical step in the emergence of simple cellular life from prebiotic chemistry, but the chemical copying of RNA templates generates product strands that contain unusual linkages. However, this research found that such RNAs can still be functional, and that diminished stability is offset by changes that render the global RNA structure relatively unchanged.
J. Sheng, L. Li, A.E. Engelhart, J. Gan, J. Wang, J.W. Szostak, “Structural insights into the effects of 2′-5′ linkages on the RNA duplex,” PNAS, v111(8), 3050 (2014).